What Every Company Needs to Know About TSCA Reviews for Industrial Biotechnology Products
Industrial platforms that rely on the use of biotechnology are regulated by the Environmental Protection Agency (EPA) under the recently updated Toxic Substances Control Act (TSCA). Over the last decade, the use of biotechnology has gone well beyond traditional enzyme and ethanol manufacture to enable a host of industrial products – butanol, succinic acid, polyhydroxyalkanoates (PHA), polylactic acid (PLA), fragrances, lubricants, and fuels. In the future, products such as cowless leather and bioluminescent microbes may be developed for consumer uses and these would be subject to EPA’s jurisdiction under TSCA.
According to a recent National Academy of Sciences (NAS) Report[1], EPA has seen an increase in the number of biotechnology products that are being submitted to it for regulatory purposes under TSCA. The number of microbial commercial activity notice submissions to EPA doubled from 2012 to 2013 and grew sharply in subsequent years.
Reprinted with permission from Preparing for Future Products of Biotechnology (2017), by the National Academy of Sciences, Courtesy of the National Academies Press, Washington, D.C.
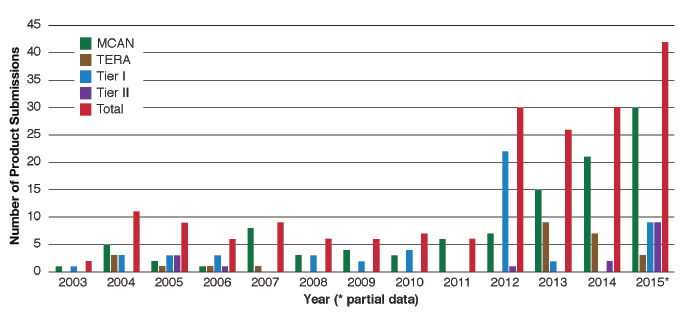
Increase in Toxic Substances Control Act (TSCA) biotechnology product submissions to the U.S. Environmental Protection Agency, 2003–2015.
NOTES: MCAN = Microbial commercial activity notice; TERA = TSCA experimental release application. A Tier I exemption requires certain certifications and recordkeeping. A Tier II exemption requires certain certifications and a notification to EPA and EPA review of specific physical containment and control technologies. SOURCE: EPA (2015).
EPA has identified that increasingly the newer submissions are from “companies that have had little or no experience with new substance review under TSCA” (EPA, 2015). Many companies with limited regulatory experience are working on biotechnology-product development. Companies with products that have not yet entered the regulatory system need to know that the federal regulatory system is use-specific and science-based. Developers of these products should identify their market and the regulatory pathway that comes with it early in the development process.
To begin to understand regulatory obligations under TSCA, companies should know that both the microorganisms that are used for production and the chemicals they produce are regulated. This is due to the way that EPA interprets the definition of the term “chemical substance” to include the microorganisms used in these processes.
The way EPA regulates chemical substances is based on their formal chemical identity and their status as “new”, “existing”, or exempt from listing on the TSCA Chemical Substances Inventory (TSCA Inventory). Section 5 of TSCA requires premanufacture notification (PMN) submissions from manufacturers and importers of microorganisms that are considered by the agency to be “new” chemical substances.[2] By statute, EPA has up to 90 days to review PMNs and MCANs but changes that Congress made to the law in 2016 now make it a requirement to have formal EPA approval in writing before a company can commercialize. The need to document these decisions, combined with increased rigor with which EPA is conducting these reviews, causes substantial delays. The agency continues to struggle with its backlog. As a means of discharging its obligations to regulate conditions of use, EPA is imposing more consent orders to regulate chemicals than ever before in the 40-year history of the TSCA program. As a result, all companies – even the most sophisticated – that engage with the TSCA program should re-evaluate their regulatory strategy.
The concepts of containment and inactivation are closely associated with EPA’s regulatory approach under TSCA. When containment and inactivation are designed into the manufacturing process it makes for more predictable regulatory outcomes. In contrast, a microorganism designed for release into the environment presents more complex regulatory challenges. Few microbes engineered for open release into the environment have been approved under TSCA although this area may be gaining momentum. Efforts have been under way for many years to genetically engineer microbes for bioremediation (Cases and de Lorenzo, 2005) and as environmental biosensors (Xu et al., 2013). Biomining and oil extraction are emerging application areas as are polymers and other chemicals that may be produced by plants for industrial use. Farther off on the horizon, EPA may see genomically recoded organisms (GROs) that are engineered to allow for tightly controlled release applications (with so-called “kill switches”). Given their increasing distance from the parental species and genus, the agency is more likely to encounter GROs initially as contained products.
What Kind of Information Does EPA Need in a TSCA Submission?
New chemical submissions for microorganisms are called Microbial Commercial Activity Notices (MCANs). A microorganism is new for purposes of TSCA if it includes a deliberate combination of genetic material that was first identified in a microorganism of a different taxonomic genus.[3] Therefore, only “intergeneric” strains require EPA review. Microorganisms that are not “intergeneric” are automatically included on the Inventory.[4] In addition, even in the case of an intergeneric strain, if the only introduced genetic material is well-characterized, non-coding regulatory regions from another genus, EPA review is not required.[5] Under TSCA, the term “microorganism” includes yeast, microalgae, bacteria and various other microorganisms, but does not include, for example, macroalgae, other plants, or cell cultures of insect, fish, or mammalian cells.[6] The information required for an MCAN includes:
- Submitter identification;
- Microorganism identity information, including a description of the recipient and new microorganisms, genetic construction, and phenotypic and ecological characteristics;
- Byproducts of manufacture, processing, use, and disposal of the new microorganism;
- Total production volume;
- Use information;
- Description of worker exposures and environmental releases; and
- Existing health and environmental effects data.
The MCAN must include a significant level of detail on identity, and the source of the parental strain should be carefully documented. A discussion on taxonomic designation should be careful to note and explain any changes in taxonomy from the published literature that could change the risk assessment. These changes in taxonomic designations happen with some frequency in the case of microalgae as science advances. Information on phenotype and genotype should identify ways in which the new microorganism can be accurately and unambiguously identified. In addition, to address EPA’s mandate to evaluate conditions of use, the submission should explain the known use or uses of the strain and offer a rationale for why other uses are not reasonably anticipated from a technical or commercial basis.
EPA’s regulations do not require that companies generate kill curve and inactivation data upfront, but providing this information in advance with the submission are advisable because the agency typically asks for this documentation during the review period. EPA will want to see a progression of data points for the time and temperature needed to inactivate the strain, as well as a resuscitation step. Risk assessment information can be provided in a robust discussion of the literature on the parental strain, together with an analysis of the effects, if any, of the modification on behavior. Manufacture and process descriptions should carefully address any release points in the process and potential exposure to susceptible subpopulations.
As for the products that are the result of these production systems, drop in replacements can have the same chemical identity as their synthetic counterparts if the chemical identity is well-defined such as ethanol, ethylene, acetate, and butanediol. On the other hand, EPA considers certain biologically-sourced chemistries such as fatty acids and polymers to be new chemical substances subject to new chemical review. In addition, when the inactivated biomass is used in TSCA-regulated applications such as fertilizers or polymer fillers these products fall under the purview of TSCA regulation. TSCA includes an exemption for the use of spent byproducts as a fuel.
Regulatory Outcomes
EPA has the authority under section 5 of TSCA to clear the planned use of an intergeneric strain without imposing regulatory controls. In addition, section 5 allows the agency to negotiate a consent order with the MCAN submitter to limit the planned use of the strain and impose operating conditions. The conditions EPA may choose to regulate include workplace protection, testing, waste disposal, and restrictions on releases to water from the facility. Typically, a consent order is followed by a proposal to impose a significant new use rule (SNUR) that includes the same restrictions to provide a level playing field for other manufacturers and importers of the notified strain, since a consent order only binds the company that negotiates and signs the order with EPA. The MCAN is also the proper form for notifying EPA of significant new uses for microorganisms subject to a SNUR.[7] Such microorganisms may or may not be intergeneric.[8]
Do I Need to Do Anything Else After EPA Finishes Reviewing My MCAN?
As with any new chemical substance reviewed by EPA, submitting an MCAN does not place the reported microorganism on the TSCA Inventory. The MCAN strain or strains will be added to the TSCA Inventory once EPA receives a valid Notice of Commencement of Manufacture or Import (NOC) from the submitter. Until then, the MCAN strain remains a new chemical substance.
NOCs need to be submitted electronically to EPA within 30 days of first non-exempt commercial manufacture or import, and must include (1) the specific chemical identity of the microorganism; (2) the MCAN number; and (3) the date when the substance was first manufactured or imported by the MCAN submitter for a non-exempt commercial purpose.[9] Your confidential business information (CBI) claims need to be reasserted and substantiated when you file the NOC. Under section 14 of TSCA, EPA must, with limited exceptions, review all substantiations for CBI claims for confidential chemical identity in every case, and such claims are only good for a period of 10 years, subject to renewal. The agency must review 25% of all other CBI claims. A discussion of the causal relationship between the disclosure of the information and competitive harm needs to be provided for the CBI claim to be upheld. The agency will notify Submitters if the information is adequate or deficient, and companies may challenge a deficiency determination by filing a request for restraining action in U.S. District Court. Companies need to take this step very seriously because the agency is not obligated to provide an opportunity to cure the deficiency outside of the formal appeals process.
Exemptions
The standard TSCA exemptions to having to make a new chemical submission apply in these cases, such as when the use of the intergeneric strain is not regulated by TSCA (e.g., pesticides, foods, drugs, and cosmetics).[10] A Test Marketing Exemption (TME) or a TSCA Experimental Release Application (TERA) may be available to conduct research and development under circumstances where the new strain is not contained.[11] In addition, there are exemptions for certain lower risk activities. The “Tier I” exemption permits manufacture or import without EPA review for a group of ten “well-characterized” strains listed in the regulations. In that case, a certification is submitted to EPA ten days before commencing manufacture or import.[12] If all conditions of the Tier I exemption are met except the specified containment and control procedures, a “Tier II” exemption notice can be filed to request approval for the alternative procedures. [13] The notice is subject to a 45-day review period. Manufacture or importation may not proceed until EPA approves the exemption when the Tier II exemption is invoked.[14]
TSCA During Research and Scale Up
TSCA requires that R&D conducted by a company that receives funds from a Federal agency requires compliance with National Institutes of Health (NIH) “Guidelines for Research Involving Recombinant DNA Molecules”. [15] Other R&D needs to be conducted in a structure, in a contained manner that meets EPA requirements found at 40 C.F.R. §§ 725.234 and 725.235. These rules require that a technically qualified individual (TQI) be designated at the site who is qualified to assess and certify that the equipment at the facility can contain the R&D strain(s). The concept of containment in these rules does not mean complete containment. The control equipment must restrict the release of the R&D strain(s) into the environment. If EPA’s rules in this area are followed no up-front notification to EPA is required for the research activity.
When environmental releases are planned or anticipated, a TERA submission is an option to consider instead of an MCAN. This is an abbreviated submission and the EPA review period is 60 days. EPA can issue a program TERA for more than one intergeneric R&D strain. The requirements and scope of the program TERA need to be negotiated on a case-by-case basis; general guidelines have not been developed. However, a programmatic approach seems ideally suited for outdoor microalgae research facilities when more than one strain is under evaluation. In 2013, EPA approved a TERA for Sapphire Energy, Inc. to test five different intergeneric strains of the photosynthetic green algae Scenedesmus dimorphus in open ponds. The field testing called for the company to evaluate the translatability of the genetically modified strains from the laboratory to an outdoor setting and characterize the potential ecological impact (dispersion and invasion) of the genetically-modified microalgae. The research findings from this effort were published in the May 2017 edition of the journal Algal Research.
Conclusions
Under TSCA, the date that first import for a non-exempt commercial purpose may legally occur is the first day after the EPA review period ends or any day thereafter.[16] That date does not have to be a moving target for companies that know what to expect and have a strategic approach mapped out in advance. Once a substance is listed on the TSCA Inventory, any person may legally manufacture or import the microorganism under TSCA for a non-exempt commercial purpose. However, genetic construct information is entitled to confidential protection in these submissions and it determines what EPA considers to be the identity of the listed strain. As a result, it is difficult to avoid an MCAN submission by relying on an existing listing for an intergeneric strain on the TSCA Inventory unless a company has sufficient knowledge of the specific technology. Certain modifications to a strain that is the subject of a prior MCAN may not make the strain new, but EPA’s agreement may be needed to support these determinations to eliminate any questions and ensure a good compliance position.
The NAS Report mentioned earlier has three key findings that are particularly relevant to EPA submissions under TSCA. First, it concluded that the endpoints of a risk assessment for future biotechnology products are not new compared with those that have been identified for existing biotechnology products, but have the potential to be more complex, more ambiguous, and less well characterized. Second, the NAS noted the need for companies to have a clear understanding of the system and principles by which the products of biotechnology processes are regulated, and this includes the removal of certain regulatory barriers to expanding sustainable products. For example, streamlining the TSCA process for drop in replacement chemistry would be a step in this direction. Third, the NAS Report calls for a single point of entry approach, by establishing a government clearinghouse function to help point companies toward the appropriate regulatory pathway, be it EPA or another federal agency. This function appears a long way off, and the trade-off could be more regulation than what is needed in some cases. There is no single regulatory pathway right now to facilitate the marketing of commodity chemical substances in the United States. This is by design. Each regulatory agency has a particularly deep understanding of the technology associated with the uses under its jurisdiction. Overall, the outlook for the growth of this sector in the NAS Report is positive and underscores the importance of understanding EPA’s role in regulating the bioeconomy under TSCA.
_____________________________________________________________
[1] Preparing for Future Products of Biotechnology, NAS 2017 https://nas-sites.org/biotech/.
[2] 62 Fed. Reg. 17,909 (Apr. 11, 1997) (codified at 40 C.F.R. Part 725).
[3] 40 C.F.R. § 725.3.
[4] 40 C.F.R. § 725.8(b).
[5] 40 C.F.R. § 725.3.
[6] See 40 C.F.R. § 725.3.
[7] 40 C.F.R. § 725.900(b).
[8] Burkholderia Cepacia Complex; Significant New Use Rule, 68 Fed. Reg. 35,315 (June 13, 2003) (codified at 40 C.F.R. § 721.1075) (for Burkholderia cepacia complex (Bcc), a group of naturally occurring microorganisms, for use other than research and development (R&D) in the degradation of chemicals via injection into subsurface groundwater).
[9] In addition, if the substance's chemical identity was claimed as CBI, and the submitter wishes to maintain confidentiality after non-exempt manufacture or import has commenced, the CBI claim must be reasserted and re-substantiated at the time of filing the NOC in accordance with 40 C.F.R. § 725.85(b).
[10] 40 C.F.R. § 725.8(c).
[11] 40 C.F.R. Part 725 Subpart F.
[12] The Tier I regulations are codified at 40 C.F.R. §§ 725.400–725.426.
[13] 40 C.F.R. § 725.428.
[14] 40 C.F.R. § 725.470(g). the journal
[15] 40 C.F.R. § 725.232.
[16] 40 C.F.R. § 725.190.

